Key Takeaways:
- Pediatric hematology is the medical specialty focused on blood disorders in children. These conditions affect red blood cells, white blood cells, platelets, and clotting factors, and they can range from mild and manageable to complex and lifelong.
- Common childhood blood disorders include anemia, sickle cell disease, hemophilia, and thrombocytopenia. Each condition has distinct symptoms, but many share warning signs like unusual bruising, fatigue, and frequent infections.
- Early diagnosis makes a meaningful difference. Many blood disorders in children are identifiable through routine blood work, newborn screening, or specialized laboratory testing.
- Treatment is highly individualized. Depending on the condition, care may include nutritional support, medication, transfusions, or advanced therapies like hydroxyurea or stem cell transplant.
- A pediatric hematologist provides specialized expertise that general pediatricians may not. Children’s blood and clotting systems differ from adults’, and accurate diagnosis requires providers trained specifically in pediatric blood disorders.
Pediatric hematology is the branch of medicine dedicated to diagnosing, treating, and managing blood disorders in infants, children, and adolescents. These disorders affect how the body produces blood cells, carries oxygen, fights infection, and controls bleeding. Because children’s bodies are still developing, blood disorders can influence growth, immunity, and overall health in ways that differ significantly from adults. A pediatric hematologist is a physician with specialized training in identifying and treating these conditions using age-appropriate diagnostic tools and therapies.
For many parents, hearing the words “blood disorder” can feel overwhelming. The good news is that pediatric hematology has advanced substantially over the past several decades. Conditions that were once poorly understood now have well-established treatment protocols, and children with blood disorders are living longer, healthier lives than ever before. This guide walks through the most common blood disorders seen in children, what symptoms to watch for, how these conditions are diagnosed, and what treatment may look like.
What Are the Most Common Blood Disorders in Children?
Blood disorders in children fall into several broad categories based on which component of the blood is affected. Some are inherited (passed down through a parent’s genes), while others are acquired through immune system changes, nutritional deficiencies, or other medical conditions.
The most common pediatric blood disorders include anemia (low red blood cells or hemoglobin), sickle cell disease, hemophilia and other bleeding disorders, thrombocytopenia (low platelet count), and clotting disorders (thrombophilia). Each of these conditions affects the blood in a different way, but they share one important feature: early recognition and specialized care can significantly improve a child’s quality of life.

What Is Anemia in Children and What Causes It?
Anemia occurs when a child’s body does not have enough healthy red blood cells to carry adequate oxygen to tissues and organs. It is one of the most frequently diagnosed blood conditions in pediatric patients.
Several types of anemia affect children. Iron-deficiency anemia is the most common, often resulting from dietary gaps or periods of rapid growth. Aplastic anemia is a rarer and more serious form in which the bone marrow does not produce enough blood cells. Hemolytic anemia occurs when red blood cells are destroyed faster than the body can replace them. Thalassemia is an inherited condition that affects hemoglobin production and varies in severity.
Parents may notice symptoms such as persistent fatigue or low energy, pale skin (especially around the eyes, nails, and lips), irritability or difficulty concentrating, shortness of breath during physical activity, and a rapid heartbeat.
A complete blood count (CBC) is typically the first step in diagnosing anemia. Depending on the type, treatment may include iron supplementation, dietary changes, blood transfusions, or, in severe cases, referral for bone marrow evaluation.
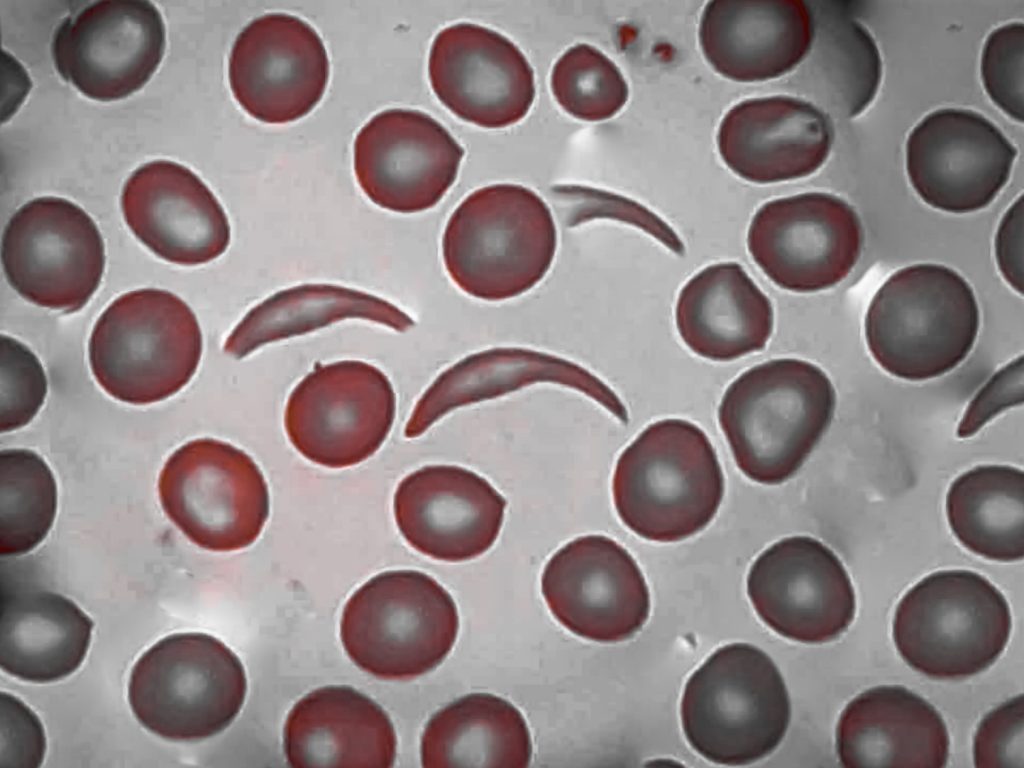
How Does Sickle Cell Disease Affect Children?
Sickle cell disease (SCD) is an inherited blood disorder in which the body produces an abnormal form of hemoglobin, the protein inside red blood cells that carries oxygen. This abnormal hemoglobin causes red blood cells to become rigid, sticky, and shaped like a crescent or sickle. These misshapen cells can block blood flow through small vessels, leading to pain, organ damage, and increased risk of infection.
SCD is typically identified through newborn screening shortly after birth. A follow-up test called hemoglobin electrophoresis confirms the diagnosis. Children inherit sickle cell disease when they receive a sickle cell gene from each parent. A child who inherits only one copy of the gene has sickle cell trait, which is not a form of the disease, though they can pass the gene to their own children.
Symptoms can appear as early as four months of age and may include pain episodes (often called pain crises) in the chest, abdomen, joints, or limbs, swelling of the hands and feet (dactylitis), frequent infections, fatigue and jaundice (yellowing of the skin or eyes), and delayed growth.
Treatment for sickle cell disease is lifelong and focuses on preventing physical and behavioral complications, managing pain, and reducing the frequency of crises. Common approaches include hydroxyurea (a daily medication that helps reduce painful episodes), regular monitoring by a pediatric hematologist, preventive antibiotics and vaccinations, blood transfusions for severe anemia or stroke prevention, and hydration and pain management plans. Stem cell transplant (bone marrow transplant) remains the only established cure for sickle cell disease, though newer gene therapies are also emerging as potential options.
What Are Hemophilia and Other Bleeding Disorders in Children?
Hemophilia is an inherited bleeding disorder caused by a deficiency in one of the proteins (called clotting factors) that help blood clot. Children with hemophilia bleed longer than normal after an injury, surgery, or even without an obvious cause. There are two primary types: Hemophilia A, caused by a deficiency of clotting factor VIII, is the more common form. Hemophilia B, caused by a deficiency of clotting factor IX, is sometimes called Christmas disease.
Von Willebrand disease is another common bleeding disorder in children. It results from a deficiency or dysfunction of von Willebrand factor, a protein that helps platelets stick together to form clots.
Warning signs that a child may have a bleeding disorder include easy or excessive bruising, prolonged bleeding from cuts, dental work, or nosebleeds, blood in the urine or stool, and in the case of hemophilia, bleeding into joints (which can cause swelling and pain).
Treatment for bleeding disorders depends on the specific condition and its severity. Factor replacement therapy is the standard treatment for hemophilia, delivering the missing clotting factor through infusion. Other medications, including antifibrinolytic drugs, can help stabilize clots. Families also work with their care team to develop preventive plans that reduce injury risk while allowing children to remain active.

What Is Thrombocytopenia in Children?
Thrombocytopenia is a condition in which a child has a lower-than-normal number of platelets, the blood cells responsible for clotting. When platelet counts are low, even minor bumps or cuts can cause disproportionate bleeding or bruising.
The most common form in children is immune thrombocytopenia (ITP), in which the immune system mistakenly attacks and destroys platelets. ITP often develops after a viral illness and, in many children, resolves on its own within several months. Other causes of low platelet counts can include bone marrow disorders, certain medications, or inherited conditions.
Symptoms parents may notice include unexplained bruising, tiny red or purple spots on the skin (called petechiae), bleeding gums, and nosebleeds that are difficult to stop.
Diagnosis typically involves a CBC and a review of the child’s medical history. In some cases, additional testing may be needed to rule out other underlying conditions. Treatment for ITP ranges from watchful waiting (when the condition is mild) to medications that help boost platelet production or modulate the immune response.
What Are Pediatric Clotting Disorders?
While bleeding disorders involve too little clotting, thrombophilia and other clotting disorders involve too much. Children with these conditions form blood clots more easily than normal, which can obstruct blood flow and damage organs.
Pediatric clotting disorders may be inherited (genetic thrombophilia) or acquired, sometimes developing after surgery, prolonged immobility, central line placement, or in association with other medical conditions. Though less common in children than in adults, clotting disorders do occur and require careful monitoring.
Signs that a child may have a clotting disorder include swelling, pain, or warmth in a limb (particularly one-sided), unexplained headaches or vision changes, shortness of breath or chest pain, and skin discoloration near the site of a clot.
Treatment typically involves anticoagulant medications (blood thinners), along with lifestyle modifications and ongoing monitoring to prevent recurrence. A pediatric hematologist works closely with the family to balance effective treatment with the realities of an active child’s life.
How Are Blood Disorders Diagnosed in Children?
Diagnosing blood disorders in children begins with recognizing symptoms and pursuing appropriate testing. Many conditions are first detected through routine blood work at a well-child visit or through newborn screening programs.
Key diagnostic tools in pediatric hematology include the complete blood count (CBC), which measures red blood cells, white blood cells, and platelets; hemoglobin electrophoresis, which identifies abnormal hemoglobin types; coagulation studies (PT and PTT), which evaluate how well the blood clots; peripheral blood smear, which allows a lab professional to examine blood cells under a microscope; and genetic testing, which can confirm inherited conditions.
Laboratory accuracy matters enormously in pediatric hematology. Blood values in children differ from adults and change as children grow, which means pediatric-specific reference ranges and experienced laboratory professionals are essential for accurate interpretation.
The laboratory teams whose precision behind the scenes directly supports every diagnosis a pediatric hematologist makes. At Cure 4 The Kids Foundation, our CAP-accredited laboratory meets the gold standard for quality, ensuring that the test results our physicians rely on are accurate, reliable, and timely.

When Should a Parent See a Pediatric Hematologist?
A referral to a pediatric hematologist may be appropriate when a child shows persistent or unexplained bruising or bleeding, chronic fatigue, paleness, or shortness of breath that doesn’t improve with standard treatment, abnormal blood counts detected on routine lab work, a family history of a known blood disorder, or symptoms that suggest a clotting event.
Pediatric hematologists are specifically trained to evaluate blood disorders in children, whose physiology, blood cell development, and clotting systems are distinct from adults. This specialized training matters because a lab result that would be normal for an adult may be abnormal for a child at a particular age, and because treatment plans must account for a child’s growth, activity level, and developmental needs.
Where Can Families in Nevada Find Pediatric Hematology Care?
Cure 4 The Kids Foundation provides comprehensive pediatric hematology services as part of its multidisciplinary care model. As Nevada’s only dedicated nonprofit pediatric cancer and rare disease treatment center, C4K offers evaluation, diagnosis, and long-term management for a wide range of non-malignant blood disorders, including anemia, sickle cell disease and other hemoglobinopathies, hemophilia and bleeding disorders, thrombocytopenia, and clotting disorders.
C4K’s team of hematology providers brings specialized expertise from nationally recognized training programs. The organization also participates in clinical research and trials, giving Nevada families access to the latest evidence-based treatment protocols. On-site services include a CAP-accredited laboratory, infusion center, physical therapy, child life specialists, social work support, and an on-site learning center in collaboration with Clark County School District so that children can continue their education during treatment.
Frequently Asked Questions About Pediatric Hematology
What is pediatric hematology?
Pediatric hematology is the medical specialty focused on diagnosing, treating, and managing blood disorders in infants, children, and adolescents. These disorders can affect red blood cells, white blood cells, platelets, and the proteins involved in blood clotting.
What are the signs of a blood disorder in a child?
Common signs include unusual or easy bruising, prolonged bleeding from cuts or nosebleeds, persistent fatigue or paleness, frequent infections, unexplained pain in the limbs or abdomen, and tiny red or purple dots on the skin (petechiae). If you notice any of these symptoms, speak with your child’s pediatrician about whether a referral to a hematologist is appropriate.
Is sickle cell disease curable?
Stem cell transplant (bone marrow transplant) is currently the only established cure for sickle cell disease. Gene therapies have received FDA approval in recent years and represent a promising new frontier. However, many children with sickle cell disease are successfully managed with medications like hydroxyurea, preventive care, and regular monitoring by a pediatric hematologist.
What is the difference between a pediatric hematologist and a regular hematologist?
A pediatric hematologist has completed additional fellowship training specifically focused on blood disorders in children. This matters because children’s blood cell development, normal lab values, and clotting systems change as they grow. Pediatric hematologists are trained to interpret results and design treatment plans within these age-specific parameters.
Does Cure 4 The Kids Foundation treat blood disorders?
Yes. Cure 4 The Kids Foundation offers comprehensive pediatric hematology services, including evaluation, diagnosis, and ongoing management of conditions such as anemia, sickle cell disease, hemophilia, thrombocytopenia, and clotting disorders. C4K’s team of hematology providers is supported by a CAP-accredited laboratory, infusion center, and wraparound support services.

About the Author: Annette Logan-Parker brings over 30 years of experience in pediatric oncology to her role as Founder and Chief Advocacy & Innovation Officer at Cure 4 The Kids Foundation. She has dedicated her career to improving outcomes for children with cancer and ensuring equitable access to cutting-edge treatments for all families.